How our earliest cells shape and maintain our health
In today’s world, we’re bombarded with information about our health: nutrition, supplements, fitness routines. We hear constantly about aging, longevity, our gut health, and inflammation. Whatever your ailment, someone has a hack for you. A compelling quick fix that promises results. These trends can feel reassuring, a reminder we’re not alone in trying to keep healthy. But maintaining health, especially as we age, is more than the latest wearable, supplement, or cold plunge. Sure, these tools may fine-tune our experiences, but beneath all the noise about optimization, there is a quieter story. One that begins at the smallest possible scale, long before we even enter the world.
Once, you were a cell. Just a single cell for a brief moment in time. No heartbeat, no brain, no fingers or toes. A single fertilized egg, a zygote, holding the instructions for all that comes next: your eyes, your bones, your knees that will heal from your first and last scrapes. This first cell is the story of where your life—and health—begins. Long before any of us knew what health was, our bodies began building, repairing, and refining themselves from this single starting point.
This first cell contains all of your possibilities, your potential. But that potential doesn’t unfold all at once. It becomes shaped, and even limited, over time. We are not developed in a single miraculous act, but rather as a line of cellular descendants from this first cell, each making commitments that will form our organ systems and lifelong health. Like branches stemming from their tree trunk, this earliest cell will give rise to all that comes next. These origins matter because these early cellular decisions ripple outward, not only shaping how our organs form but how our bodies will repair themselves decades later. This very first cell, a progenitor, contains all of our potential. But what follows depends on timing, environment, and even chance.
The Ultimate Progenitor & A Biological Hierarchy
By the dictionary, a progenitor is defined as ‘a person or thing from which another originates.’ An ancestor, a parent, a matriarch. This same process applies to cell biology. A progenitor is a parent cell, one that can divide to make new cells, which we call daughter cells. This one cell becomes two, two become four, four become eight, and so on. From that single cell, over the course of nine months the human body will build itself, generating some 30 billion cells by birth, each with a role to play.
But simply dividing into new cells isn’t enough. Very early on, tiny cellular decisions will decide the fate of these daughter cells. Some will commit to becoming neurons, wiring the brain and spinal cord, others will form the liver, kidneys, your fingers and toes. These cells will eventually branch out to become the more than 200 different cell types in your body. Scientists call this process of cellular specialization differentiation.
With this commitment, as cells are set on a path to specialize into one cell type, they lose their ability to become other cell types. Our neurons cannot then transform into liver cells. And as cells differentiate or mature into fully functioning neurons or liver cells, they will lose their ability to divide altogether. Our cells become incredibly proficient at doing one job, and incapable of doing most others. A biological hierarchy is now in place.
In biology, we more commonly refer to progenitor cells as stem cells. As the stem from which all other cell types branch off. This term can carry a lot of baggage. In the public space, stem cells are almost exclusively associated with ethical debates over the use of embryos or embryonic stem cells in scientific research. But in scientific circles, the term ‘stem cell’ can spark a different kind of debate. These disagreements center more on what defines a true stem cell. How far down the branches of our biological hierarchy still qualify as a ‘stem’? Some argue it is only those at the very top, while others argue that all cells capable of creating new daughter cells or branching further. Scientists can also disagree on how do we define each of these cell types. What characteristics to they display that definitively indicates a specific type of stem cell.
These are the types of scientific nuances you won’t read about in textbooks, or even very often in scientific research. They are often reserved for scientific spaces: peer review, scientific conferences, or if consensus has been reached, a position statement from a research society. This is one reason I like the term progenitor. Largely free from debate, it simply defines all cells that can divide to make new cells.
Progenitors in Early Life
At the very top of our biological hierarchy sits our single fertilized egg, the zygote. The full potential that these cells contain is referred to as referred to as totipotency—from toti meaning total, and potens meaning power or ability. While this cell will divide rapidly, the progeny are limited in their ability to grow in size, both by the constraints of the embryo size and also by their biological cues. Within a few days, they will have moved down the hierarchy, no longer carrying totipotency.
Next, sit the embryonic stem cells. They can divide again and again, rapidly producing the many cells needed to build a developing fetus. While not totipotent, these cells still carry remarkable potential, alongside nearly unrestricted cell division. These features make these cells incredibly powerful for research and potential therapies. No longer constrained from growing, embryonic stem cells also carry the ability to become almost any cell type in the body: pluripotency—from pluri meaning many. These features are also temporary, however, gradually lost as cells commit to their more specialized roles.
The first branches to emerge from the embryonic stem cell trunk are three foundational groups of cells known as the germ layers. These layers represent the earliest commitments in development, narrowing the future paths of these cells and their daughter cells. It is notable that these germ layers were first described in the early nineteenth century. Rudimentary by today’s technology, early embryologists used simple microscopes with natural light to study this development in chicken eggs. Prior to this, one theory of development posited that tiny versions of the animals developed fully formed inside the eggs. This early work visualizing layers of cells developing over time would lay that theory to rest.
By the mid-1800’s, scientists recognized that most animal embryos organized into two or three layers of these cells. They called them germ layers, which reflects how early embryologists understood them: as layers that would sprout new systems, like a germinating seed. Like the main limbs of a tree, these layers do not build the body directly, but give rise to everything that will. But the significance of these germ layers within our biological hierarchy of cells would not become clear until until one scientist, Robert Remak, began to connect the dots.
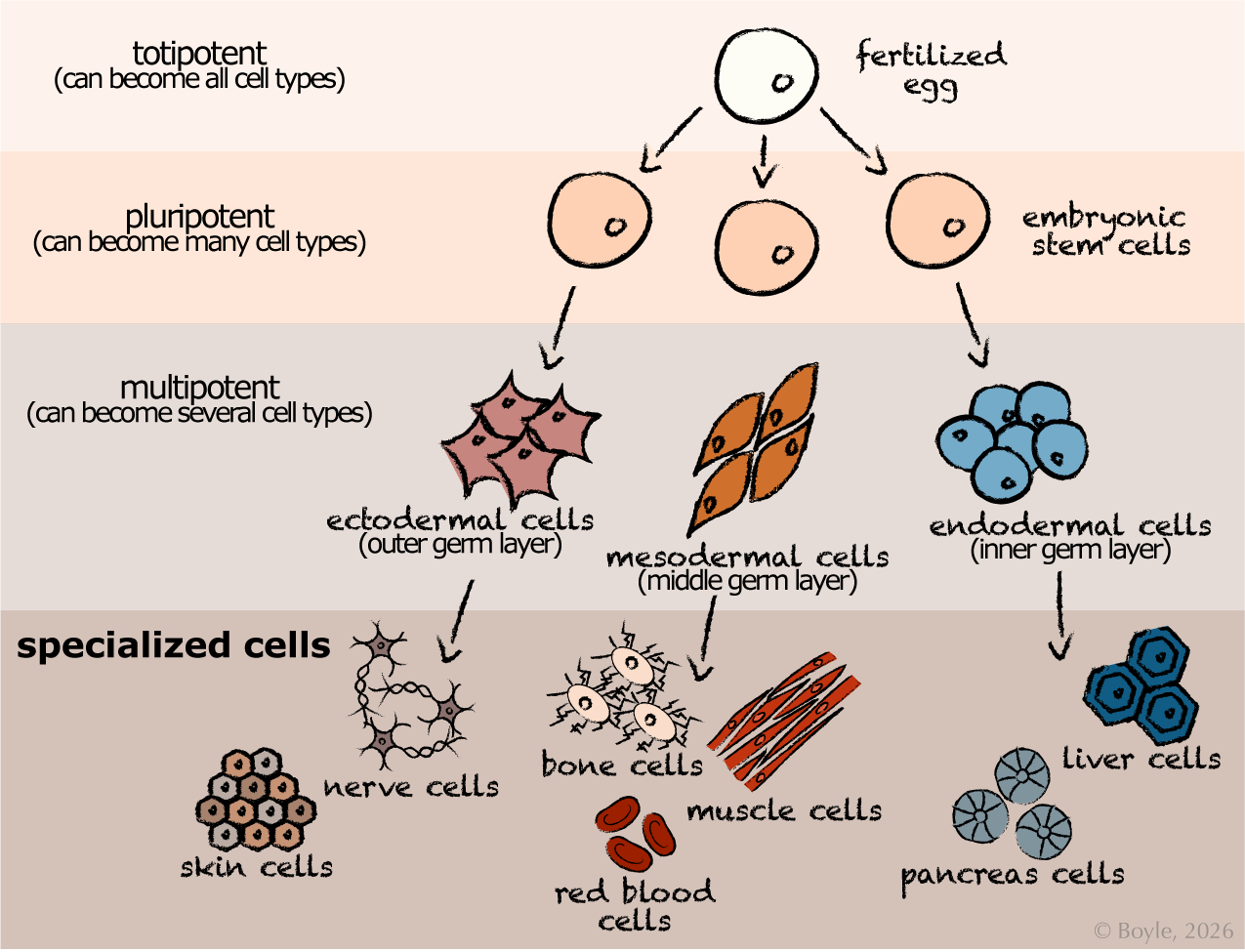
This figure shows the hierarchy of cell potency from the fertilized egg to specialized cell types. This hierarchy illustrates the progressive narrowing of cellular potential during early development alongside the widening breadth of cell types developed. The fertilized egg begins as a totipotent cell, capable of giving rise to all cell types. As development proceeds, embryonic stem cells become pluripotent, able to form many cell types. These then give rise to the three multipotent germ layers: ectoderm, mesoderm, and endoderm. Each germ layer produces more restricted progenitor cells that develop into specialized cell types and tissues, including skin, nerve, bone, muscle, red blood, liver, and pancreas cells.
Remak was a careful scientist and skilled microscopist. He made several important discoveries in his time but one thing he did exceptionally well was to pull it all together. Based on his observations and the work of others, he had an impressive ability to to synthesize information and create new theories. In the 1840-50’s, he had made the case that these germ layers were not simply developmental structures, but the source of every organ. That each tissue could trace its roots back to one of these three layers. That an organized hierarchy was in place.
The cells of the germ layers are still considered stem cells within the embryo, but they are no longer embryonic stem cells. They have now become multipotent, capable of producing only a limited number of cell types. Remak defined three germ layers based on their location in the embryo and the organs they would produce. First is the ectoderm, the outermost layer that produces the parts that will interface with the outside world: skin and hair, tooth enamel, and the nervous system, including the brain and spinal cord. Beneath it lies the mesoderm, the middle layer that forms the body’s structural and transport systems: bone, muscle and connective tissues, as well as the kidneys, heart, and cardiovascular system. The innermost layer, the endoderm, will become the gut and gastrointestinal tract, lungs and trachea, as well as important organs such as liver, pancreas, and thyroid.

The mesoderm, remarkably, will branch yet again. Some of its cells will form what is called the extra-embryonic mesoderm, that portion that exists outside of the embryo itself. These cells will not become part of the fetus. Instead, they construct the placenta and umbilical cord, the temporary but essential organs that sustain the fetus as it grows.
Proper development of all three germ layers is critical for health. Complications in these processes early in development can lead to severe, often fatal consequences. For example, disruptions in the ectoderm can contribute to neural tube defects, while disruptions in the mesoderm can lead to structural heart defects or certain muscular dystrophies.
From this point forward, each germ layer follows its own developmental path, committing to specific futures. Their cells will continue to divide, generation after generation, with daughter cells becoming progressively more specialized as they assemble the organs and tissues of the fetus. But once these cells reach full specialization, once they are fully differentiated into their final identities, they can no longer divide. At that point, they are no longer progenitors.
Robert Remak: Overlooked Architect of Cell Theory
Born in 1815 in Prussian Berlin, Remak made several important contributions to embryology and cell biology in his lifetime. What’s remarkable is that he accomplished much of this work on his own, without the support of academic laboratories or institutional funding. Despite being an exceptional student, graduating cum laude with a doctorate of medicine and surgery in 1838 from Johannes Müller’s prestigious laboratory, he was unable to secure a faculty position. At a time when many Jewish scientists and professionals were pressured to denounce their religion to gain access to academic jobs, Remak refused. Instead, he funded his research through earnings in his clinical practice. Remak is often reduced to the footnotes of science history. His contributions overshadowed or untold. Some even suggest part of work was plagiarized by a colleague.1
I include Remak’s story as a reminder that science has never been equally accessible to all. It is shaped by those who hold resources and power, lines that have long-aligned with whiteness, maleness, and privilege. Even today, many scientists must sacrifice more than others to enable their work. The perseverance of those who work harder, surrender more, receive less, and continue anyway is as inspiring as it is heartbreaking.
The Long Work of Progenitors
Development is only the beginning of the story. For the rest of our lives, our tissues depend on new generations of cells to keep us alive and well. The word progenitor describes much more than embryonic stem cells or only those stem cells closest to the trunk of our hierarchy. Progenitors represent the broader family of cells that not only build our tissues but shape how our bodies grow, adapt, and repair for eight decades or more.
All of our specialized cells, once differentiated, have a finite lifespan. But those lifespans vary dramatically. Some cells are built to last. Neurons, for example, can survive for an entire lifetime: eighty years or more. Female mammals, including humans, are born with all the eggs they will ever have. These cells can survive for fifty years or more. Heart muscle cells are replaced quite slowly, at a rate of about one percent per year. Meaning that by age fifty, about half of your heart cells are the same ones you were born with, and some will go on to last for your entire lifetime.
Many of our tissues contain not only the mature, fully functioning cells that carry out their daily work, but also house small populations of progenitor cells that sit quietly in reserve. In the heart, for example, these are known as cardiac progenitor cells. They lie further down the branches of our cellular hierarchy than their germ layer parents, but they still retain the capacity to divide and differentiate into new heart muscle cells. In organs with slower cell turnover, like the heart, these progenitor will reside in a state of suspended animation known as quiescence, sometimes for decades. But when maintenance is needed to replace aging or damaged cells, they exit that dormant phase and begin dividing again, creating new daughter cells that will replace the old with new.
“Early cellular decisions ripple outward, not only shaping our organs, but how our bodies will repair themselves decades later.”
At the opposite end of the spectrum are cells designed for rapid response. White blood cells, mobilized during infection or illness, live only hours to days. Red blood cells circulate for roughly four months before being replaced with newer versions. Some progenitor cells are uniquely mobile with remarkable homing capacity. The progenitors for our blood and immune cells, the hematopoietic stem cells, are a prime example. These cellular nomads primarily reside in the bone marrow, but small numbers routinely circulate through our blood and lymph nodes. They serve as sentinels for incoming infections and travel to distant organs where they maintain tissue and immune health. When injury or illness occurs, these progenitors respond to chemical stress signals released by affected organs, traveling through the circulation to where they’re needed most.
Other cells take the middle road, neither built to last a lifetime nor destined for rapid turnover. Bone is a good example. During childhood, bone is remarkably dynamic, turning over at rates approaching fifty percent each year as the skeleton grows. In adulthood, that pace slows to roughly ten percent each year, gradually replacing all the cells of your skeleton over the course of a decade. Muscle follows a different pattern, turning over quite slowly under ordinary conditions, but incredibly responsive to damage. If you’ve ever felt sore after a particularly aggressive workout, you’ve experienced this process firsthand. Muscle soreness is caused by micro-tears in your muscle cells: small injuries that trigger a rapid repair response. Within a day or two, those damaged cells are repaired or replaced by the resident progenitor cells, satellite cells, leaving the tissue stronger and better adapted to your new exercise routine.
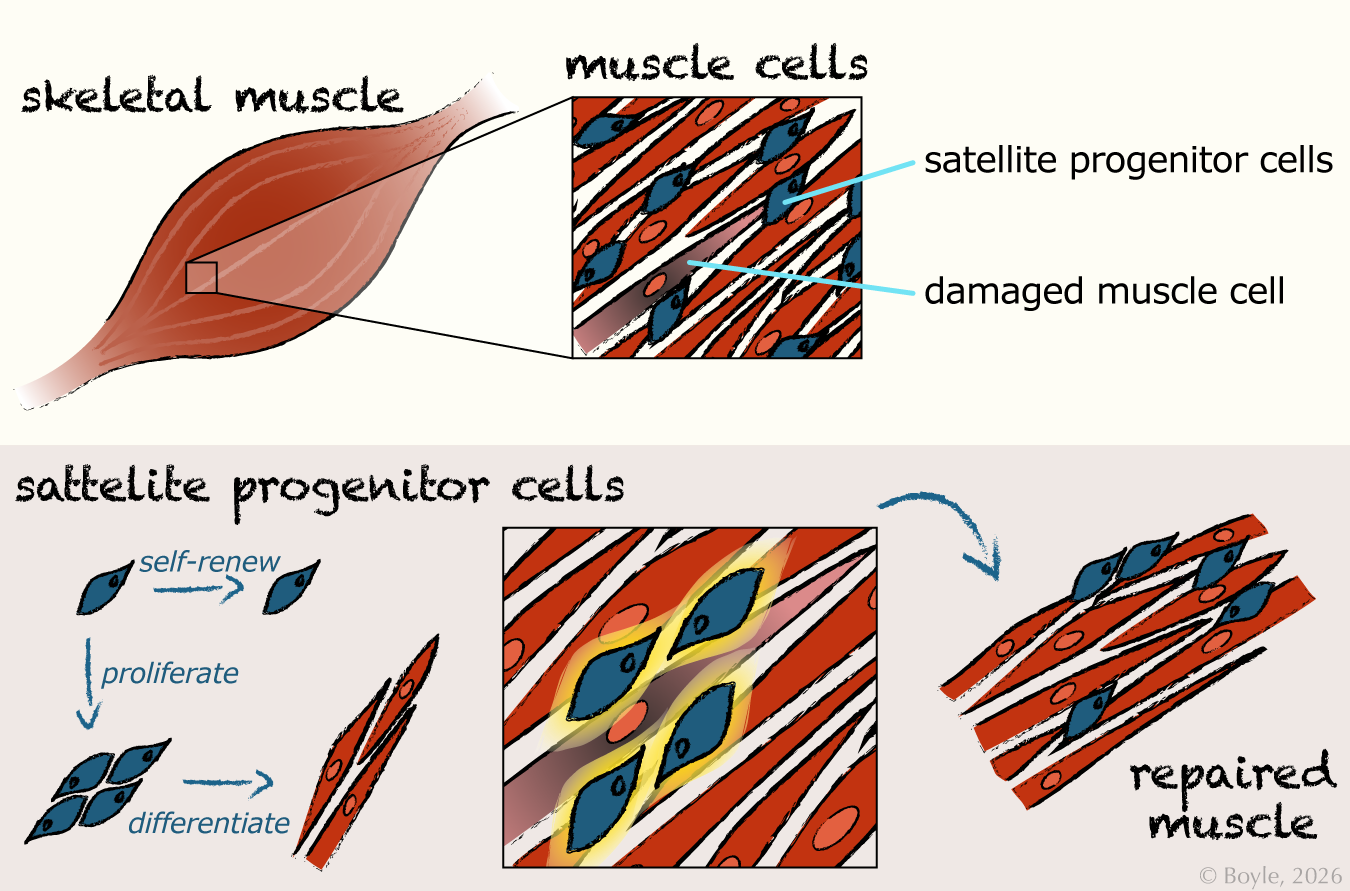
This figure illustrates how satellite progenitor cells support muscle maintenance and repair. The top panel shows a section of skeletal muscle with a zoomed‑in view highlighting elongated muscle fibers and the location of satellite progenitor cells adjacent to a damaged muscle cell. The lower panel depicts satellite progenitor cells that can undergo self‑renewal to maintain their own populations, or proliferation and differentiation to create new muscle cells. These daughter cells integrate into the injured region, restoring the structure of the muscle tissue.
This cell renewal does not happen in waves. It is a continuous process. At any given moment, our bodies are comprised of cells of many different ages. A rich tapestry of young and old. Life at the cellular level is a careful balance between cell renewal and persistence. All made possible by progenitor cells. From our early origins as a single progenitor to our deaths some eighty years later, the progeny of that cell keeps our bodies healthy and strong. How well these systems work varies from person to person and across the lifespan. Our genetics, our environment, our history, and even our age can all shape how these quiet repair systems perform over time.
The Promise of Progenitors
Subtle differences in how our progenitors build and repair our bodies are what shape our lifelong vulnerabilities and our resilience. Their role in development and maintenance of our tissues across our lifespan is remarkable, but the question that drives my research is what happens when things go awry. Consider an early disruption: a slight change to a progenitor population during fetal development. The immediate consequence could impact organ function. A lasting consequence could impact repair and renewal. Tiny adaptations that can echo for a lifetime.
In my lab, we study how small shifts in fetal stem cell metabolism and function can shape a person’s risk for obesity or diabetes years later. We use cells collected from infants’ umbilical cord after birth to understand how exposures during this early embryonic and fetal development may shift these systems, and to identify early predictors of future metabolic health. By understanding when and how progenitor cells enhance or diminish resilience, we hope to support healthier trajectories across the lifespan. Not by predicting specific futures, but by recognizing the earliest places where change is possible. Where we will find our greatest potential.
Linked References
- Figlio, K.; Weindling, P. (June 1984). “Was social medicine revolutionary? Rudolf Virchow and the revolutions of 1848”. The Society for the Social History of Medicine Bulletin. 34: 10–18. ISSN 0307-6792. PMID 11611569.


