A look at how our muscles adapt to dietary fat, and how that varies from person to person
A wise colleague once told me that, as scientists, our research products weren’t truly finished until we’ve written a summary for non-scientists. We are prolific writers. The currency of our careers is publications and grants. To date I have more than 50 publications that many of you have never read. We don’t publish these in newspapers or popular science magazines, they are tucked away in science journals meant primarily for the eyes of other scientists. Let’s change that. This is a series I might call From the Paper. A real world, explainer of my science publications.
To start, we must start at the beginning. We’re going all the way back to my graduate training, back to 2007. It’s a hot, humid summer in Greenville, North Carolina and I’m leading my first ever clinical research study: investigating how a short-term, high fat diet impacts people’s muscle metabolism. Let’s discuss my first paper,1 link here.
Reader’s Note: Here, I use the term “people with obesity” to reflect that obesity as a complex, chronic disease. The findings discussed describe group‑level patterns, not individual predictions, as does the clinical designation of obesity by BMI. People with obesity are metabolically diverse, and my ongoing research focuses on understanding this heterogeneity and its origins.
The Background
Our skeletal muscle works hard for us. It gets us around, fuels our sports, and helps us stand and even breathe. To fuel this work, muscle uses the foods we eat. Even at rest, it accounts for about 30% of our energy use. Of the major macronutrients (fats, carbohydrates, and proteins) muscle mainly uses fats and glucose for energy, while protein is used mainly for building and repairing muscle.
So how does muscle decide when to use fat versus glucose? I like to think of it as a question of how fast we need energy. Fats are energy‑dense but slow to break down, making them ideal for rest or lower‑intensity movement. Running from a tiger and need a lot of energy really fast? Glucose is your champ. Switching between these fuels, called metabolic flexibility, is regulated at many levels: in the whole body through hormones like insulin, in how tissues take up different fuels, and through cellular systems that determine what gets used and what gets stored for later.
In people with obesity (BMI > 30) and insulin resistance, this flexibility is often blunted. At this time studies showed that, compared with people of normal weight (BMI < 25), those with obesity used less fat for energy overall. And when given insulin, normal weight individuals smoothly shifted to using more glucose, while people with obesity showed little to no shift at all.2 Later work suggested this rigidity is tied to insulin resistance, common in obesity, rather than an inability of the muscle itself to switch fuels.3
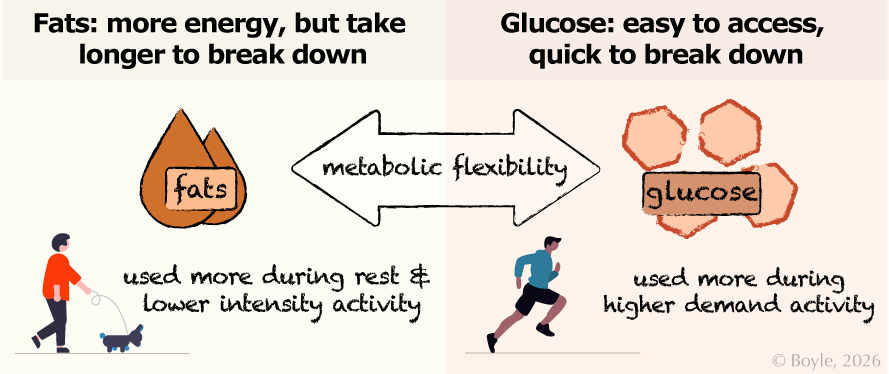
Our muscle uses both fats and glucose for energy, shifting between them depending on how quickly fuel is needed, but also other factors, like how much fuel is available. Fats store a lot more energy, meaning a single molecule contains a lot of calories. But fats take longer to break down so are used mainly during rest or low‑intensity activity. Glucose is less energy-dense, but is easy to access and supports higher‑demand activities. The ability to switch flexibly between these fuels, metabolic flexibility, is a key feature of healthy muscle metabolism.
The Question:
Most earlier work looked at how muscle responds to insulin or carbohydrates, but far fewer studies asked how people respond to eating more fat, or what actually changes inside muscle cells when fat intake rises. This study was designed to fill those gaps. Put simply, we asked:
How does muscle from people with normal weight or obesity adapt to a short high‑fat diet?
What we did:
We fed a controlled 65%‑fat diet for five days to two groups of young adults: one group of people with obesity and one with normal weight. Muscle biopsies were collected before and after a single high‑fat meal on the first and last day. In the muscle, we measured mRNA content for genes involved in fat oxidation and acylcarnitine levels. mRNA is a snapshot of which genes are active, helping us see how muscle responds at the molecular level, while acylcarnitines are by‑products of fat breakdown that show how well fat oxidation is working. Our goal was to capture very early molecular signals, within hours or days, of how muscle responds to increased dietary fat.
What we found:
mRNA responses
After a single high‑fat meal, the normal‑weight group showed large increases in PPARα, a key regulator of fat oxidation. Those with obesity showed no change. After five days of the diet, the normal‑weight group up‑regulated multiple genes involved in oxidative metabolism (PDK4, UCP3, PPARα, PGC‑1α), with PPARα increasing more than ten-fold! Again, the group with obesity showed little to no response.
Acylcarnitine responses
After five days, acylcarnitines in the normal‑weight group stayed mostly stable. But in people with obesity, they increased, especially medium‑chain acylcarnitines, suggesting fat breakdown had started but not finished, leading to a buildup of intermediates.
In short, just five days of a high‑fat diet revealed markedly different molecular responses in the skeletal muscle. These patterns reflect metabolic flexibility in the normal‑weight group: muscle sensing more dietary fat and shifting toward using more of it. In obesity, this response was severely blunted: metabolic inflexibility.

Study overview. Participants consumed a high‑fat diet (65% fat) for five days. Muscle biopsies were analyzed for expression of key fat‑oxidation genes (PPARα, PDK4, UCP3, and PGC‑1α). In individuals with normal weight (BMI < 25), these genes increased markedly after both a single high‑fat meal and after five days of high‑fat feeding, indicating greater metabolic flexibility. In individuals with obesity (BMI > 30), gene expression showed little to no change. Medium‑chain acylcarnitines increased only in the obesity group, suggesting incomplete fat oxidation.
What it means:
If skeletal muscle can’t adjust to changes in dietary fat, several downstream issues can arise. For example, a buildup of lipid intermediates such as acylcarnitines can ultimately reduce mitochondrial efficiency, and excess fat in muscle is commonly associated with insulin resistance. The early molecular differences we observed may help explain why people with obesity often show lower fat oxidation and reduced metabolic flexibility, and why some struggle more with high‑fat diets or are more prone to metabolic disease.
What we still don’t know:
This study measured molecular signals, not whole‑body fat oxidation or long‑term outcomes. We still did not know how long these changes would persist, whether they directly relate to muscle fat uptake or insulin resistance, or whether weight loss, exercise, or diet could reverse these patterns. Since I graduated, researchers from my mentor’s lab have addressed some of these questions. You can read about how exercise training can shift these patterns in people with obesity here.4 But an even bigger question remains:
Why do some people adapt easily to increased fat intake while others show these early signs of metabolic strain?
This is a question that has driven much of my work since this first study, and one we’ll continue to explore in the coming papers.
Linked References
- Boyle KE, Canham JP, Consitt LA, Zheng D, Koves TR, Gavin TP, Holbert D, Neufer PD, Ilkayeva O, Muoio DM, Houmard JA. A high-fat diet elicits differential responses in genes coordinating oxidative metabolism in skeletal muscle of lean and obese individuals. J Clin Endocrinol Metab. 2011 Mar;96(3):775-81. doi: 10.1210/jc.2010-2253. Epub 2010 Dec 29. PMID: 21190973.
- Kelley DE, Goodpaster B, Wing RR, Simoneau JA. Skeletal muscle fatty acid metabolism in association with insulin resistance, obesity, and weight loss. Am J Physiol. 1999 Dec;277(6):E1130-41. doi: 10.1152/ajpendo.1999.277.6.E1130. PMID: 10600804.
- Galgani JE, Heilbronn LK, Azuma K, Kelley DE, Albu JB, Pi-Sunyer X, Smith SR, Ravussin E; Look AHEAD Adipose Research Group. Metabolic flexibility in response to glucose is not impaired in people with type 2 diabetes after controlling for glucose disposal rate. Diabetes. 2008 Apr;57(4):841-5. doi: 10.2337/db08-0043. Epub 2008 Feb 19. PMID: 18285553.
- Battaglia GM, Zheng D, Hickner RC, Houmard JA. Effect of exercise training on metabolic flexibility in response to a high-fat diet in obese individuals. Am J Physiol Endocrinol Metab. 2012 Dec 15;303(12):E1440-5. doi: 10.1152/ajpendo.00355.2012. Epub 2012 Oct 9. PMID: 23047988.

