How science helped us understand the invisible
Vaccines, pasteurized dairy, even basic food safety. These felt like settled facts. Scientific discoveries that had moved from the lab and firmly into our daily lives. And yet, much of what we now take as obvious about disease, decay, and health was anything but clear for most of human history. This isn’t the first time science and society have struggled to reach an agreement on these concepts many of us had taken for granted. For centuries, people debated the ideas that underlie these same ‘settled’ truths, tracing back to a much simpler, but far more profound question: where does life comes from?
What feels new today is not the uncertainty itself, but the return of it. In recent years, questions many considered resolved have resurfaced, often in public and highly visible ways. It turns out that ‘settled’ may be more fragile than we assumed. And perhaps it’s time to revisit the history that resolved these debates the first time around. Not through a single decision of convenience or ideology, but grounded in scientific insights developed over centuries. A story that stretches back thousands of years, to Aristotle in 350 BC.
When life was unseen
We’ve all seen it: the forgotten heel of bread in the back of the pantry, green with mold. Fruit flies congregating around the bananas left too long. Food has been going bad since humans began storing food. While our ancestors could plainly see mammals giving birth or chicks hatching from eggs, and understood that some living things made baby living things, this understanding only extended to what the eye could see. Mold spores and bacteria were too small to see and seemingly appeared out of nowhere. Aristotle called this spontaneous generation. He posited that new life sprang fully formed from non-living matter. This theory extended to beings much larger than microbes. To Aristotle, a shrewd observer, maggots spontaneously formed on rotting meat, mud produced frogs, and slime created oysters. Without looking deeper, in many cases without the tools to look deeper, this was a very logical conclusion. In later years, some even applied this term to mice ‘generating spontaneously’ in piles of old rags. It sounds outrageous today, but the theory was quite plausible given the tools of the time and was commonly accepted for centuries.
The idea that life could arise spontaneously wasn’t just a scientific theory, it shaped how people understood decay, disease, and even the divine. Without understanding how food spoiled, there was little ability to manage its storage and keep it safe. Contamination and illness often appeared random, unpredictable. Many diseases were thought to arise from ‘bad air,’ based on miasma theory. The stench of rotting food, and in some cases bodies, was believed to carry diseases like cholera or the plague (Steven Johnson’s book, The Ghost Map, is an excellent telling of how these ideas played out during London’s 1854 cholera outbreak).
What’s intriguing is that long before modern germ theory, many people had correctly intuited that something in the air could spread illness. While not all diseases are airborne, many do travel through respiratory droplets when we cough. And even infections transmitted by other routes can quickly move through populations by human contact. These people were not naive: they saw the effects and patterns clearly, even if the underlying cause was unclear.
That same uncertainty shaped how people approached food safety. Ancient preservation techniques like drying, salting, or pickling were widely used, but most fresh foods still spoiled quickly. People learned to rely on what they could see, smell, or touch to determine what was safe to eat. Mold was obvious: fuzzy, green, clearly visible. But bacteria were far harder to recognize, and far more dangerous. Foods could look, smell, and even taste normal while carrying harmful bacteria or disease. And even what we would now consider mild food poisoning could be deadly without the means to treat it.
In short, illness felt unpredictable. A harmless meal one day could be deadly the next. One person might fall ill while the others remained well. For many, this unpredictability suggested something beyond human control, something divinely ordained. People weren’t wrong to sense that something unseen was at work. But what cannot be seen is difficult to control. So dangers were managed through experience, intuition, and belief rather than by established science. This dichotomy between what could be observed and what remained invisible shaped how people understood food, illness, and even life itself.
Disputing the Theory
By the 17th century, several scientists had begun to question spontaneous generation. People had long observed that meat left out in the open rotted and appeared to grow maggots. But while covered meat still rotted, it did not grow maggots. Moving from observation and into experimentation, the Italian physician and biologist Francesco Redi set out to systematically test the theory.1 In truth, he was looking to confirm spontaneous generation, not refute it. But try as he might to produce baby snakes from decaying snake meat, all he produced were flies. Why would it be that flies spontaneously generated from snakes, rather than more snakes?
So, Redi decided to test this more systematically. He placed various meats and non-food objects in separate containers. Some of these jars he would seal with cork, others he covered with a thin gauze, and some he left to the open air. Days later he would return to see what had appeared. Like many before him, Redi found that all of the meat decayed. But while maggots appeared in the open jars, the cork-sealed jars contained none. And yet proponents of spontaneous generation argued that this proved nothing. They claimed that sealing the jars blocked the ‘vital air’ thought to carry the life force that made spontaneous generation possible. This was a serious scientific and public debate that had already persisted for centuries. This may feel familiar even today: when key variables are beyond direct observation, disagreements not only center on conclusions, but also on what counts as a valid experiment.
What set Redi’s experiments apart from all that came before was the gauze. In these jars, the ‘vital air’ could pass freely in and out, but flies could not touch the meat. In this case, maggots appeared on the gauze, not on the meat inside. Sure, the meat still decayed. But without physical contact, life did not appear. It turns out, flies had landed on the gauze and laid eggs there. At first these eggs were not visible to Redi, but the resulting maggots were. He collected them for further study and found that these maggots eventually developed into flies.
With these experiments, Redi demonstrated that ‘vital air’ alone was insufficient to generate life, and that maggots were the offspring of flies. These were the first inklings that organisms previously thought to arise spontaneously were, in fact, from another living being. These experiments are also early lessons in how we can study things beyond direct observation, by designing experiments that expose its effects. Furthermore, these novel methods can reveal important new insights. Redi did not set out to discover that maggots came from flies. Some of the most interesting scientific discoveries are unplanned and unexpected.
While Redi’s work is often highlighted as a turning point, he was not alone in questioning spontaneous generation. Many had begun to notice similar that specific conditions were necessary to allow, or prevent, spontaneous generation. Despite these observations, the scientific and public debate over spontaneous generation would persist for another 200 years, exhaustively.
By the 1800’s, most experimentation had shifted from rotting meat and flies to meat broths that could be sterilized by boiling. When exposed to air, these broths often became cloudy: contaminated with bacteria. Yet the results from these experiments were frustratingly inconsistent. Some broths spoiled quickly, others only after long delays or not at all. For proponents of spontaneous generation, this variability reinforced their claims. They argued that boiling destroyed the ‘life-force’ in the air that was necessary for new life to form. People on both sides of the debate could clearly see the consequences, but neither could see the cause.
“The idea that life could arise spontaneously wasn’t just a scientific theory, it shaped how people understood decay, disease, and even the divine.”
In 1860, the Paris Academy of Sciences offered a prize to the person would could bring an end to the debate. This effort was backed by the Catholic Church and government appointees who opposed spontaneous generation as counter the concepts of the divine creation of life.2 They were eager to refute Aristotle’s Theory once and for all.
Enter Louis Pasteur. More famously known for patenting his process of sterilization through boiling. Against the advice of his colleagues, Pasteur took on this challenge. After all, scientists had been failing at this for hundreds of years and they did not want to see him stuck on this for the rest of his career. But Pasteur had some new ideas.
Up to this point, Pasteur’s work had largely focused on fermentation and preservation. He had already identified yeast as the critical component for fermenting beer and wine, and through these experiments, he had learned that fermentation not only involved yeast, but that yeast was a living organism completing a biological process.3 It was consuming the sugars and producing alcohol, not simply participating in a chemical conversion. Pasteur had observed that wine would not ferment if it were boiled beforehand. The boiling was killing the yeast, halting fermentation.
More than understanding how wine was made, Pasteur was deeply interested in what made wine spoil. In his earlier fermentation experiments, he had already concluded that yeasts were living organisms that came from the environment rather than generating in the wine itself. This understanding that microscopic, living organisms were floating around in the air and capable of affecting the wine, changed how he thought about spoilage altogether.
By heating beer or wine to carefully controlled temperatures, typically between 60-100°C (~140-212°F), Pasteur found he could eliminate any potential microorganisms, just as he had done with the yeast. In doing so, he preserved flavor and prevented spoilage: pasteurization. This discovery was a huge boon to wine and beer producers, who could now store their products longer and ship them further.
Through this work, Pasteur also observed that spoilage was not always the same. Some wines turned to vinegar, others became cloudy or lost their alcohol content. He spent a lot of time understanding the different conditions that would lead to each outcome. Examining the wines under a microscope he observed that the best, unspoiled wines contained mostly round microorganisms: yeasts. Spoiled wines contained a variety of other organisms: rods, spirals, chains, and each type of spoilage had different combinations of organisms.
The invisible work in your kitchen

We still rely on these principles Pasteur demonstrated every day. If you’ve ever made your own sourdough starter on the counter from “wild yeast,” you’ve already seen the process that Pasteur was studying. Fermentation of wine begins with microscopic yeast floating in the air. These organisms are far too small to see with the naked eye, yeast is a single‑celled microscopic fungus. The baker’s yeast we buy at the store is only visible because it has been dehydrated into large clumps.
If you’ve ever baked with baker’s yeast, you may also have discovered, perhaps unintentionally, how sensitive this process is. Heat your water or milk a bit too much, and the dough won’t rise. You’ve killed the yeast, just as Pasteur observed in his experiments.
If you’ve ever made jam or other canned goods, you have also participated in a process similar to Pasteur’s pasteurization. Boiling or pressurizing the jars doesn’t just seal them, it destroys the bacteria and mold spores that cause spoilage. Without those organisms, and without a way for new ones to enter, food can be stored safely for long periods. Temperature and timing matter here: boiling for long time or at high temperatures can certainly ensure you’ve killed all the microorganisms, but it can also destroy your flavor, texture, and quality. Effective preservation depends on finding a balance, the same principle Pasteur identified.
Investigating the culprit
What does all of this have to do with spontaneous generation? Because what Pasteur had discovered was the cause of the spoilage: other living organisms. Ones we couldn’t see with the naked eye. When Pasteur applied this knowledge to investigating spontaneous generation, his approach was fundamentally different from those before him. Rather than focusing on the broth or wine that spoiled, he instead focused on what was doing the spoiling. He focused on the airborne particles, the microorganisms he hypothesized were responsible. If airborne microorganisms were to blame, then preventing airborne particles from reaching the broth should prevent spoiling altogether.
Many had tried this before: put a lid on it and the broth doesn’t spoil. Pasteur, however, had the proponents of spontaneous generation to contend with. Those who argued that lids prevented the entry of “vital air,” which they believed was necessary for spontaneous life to arise. Pasteur’s answer to this was to create a series of flasks with elaborate swan-shaped necks. These would allow some air flow into the sterilized broth, while reducing or eliminating the flow of dust and other particles into the broth.
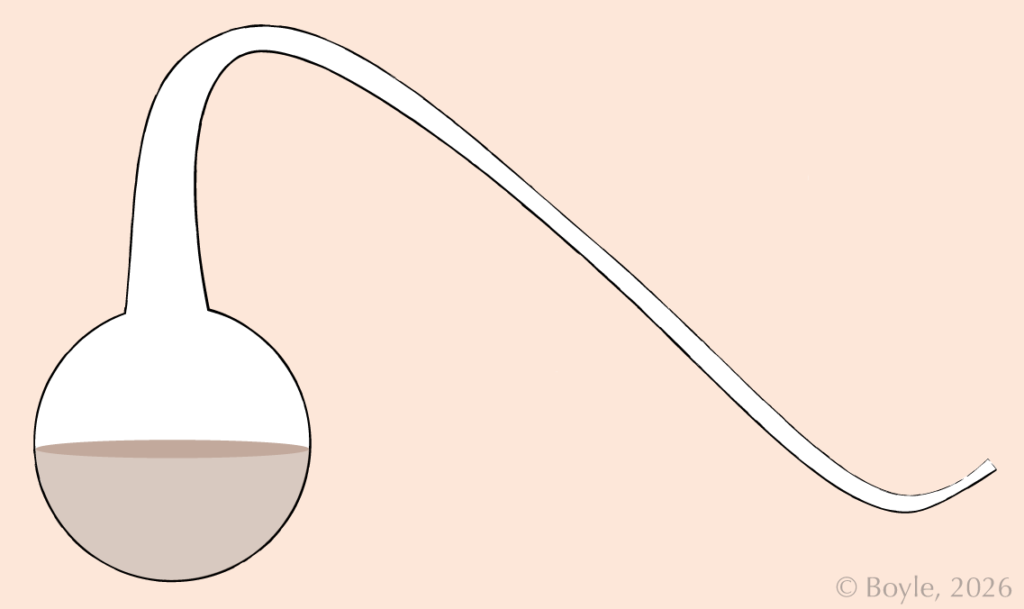
A swan‑neck flask used in Pasteur’s experiments. The curved neck allows air to enter while trapping dust and airborne particles that carry microorganisms. While the design is often attributed to Pasteur alone, some suggest these flask experiments were developed in collaboration with chemist Antoine‑Jérôme Balard.4
But Pasteur also considered another variable. Why had all before him who attempted similar experiments, albeit without swan necks, have such inconsistent results? For hundreds of years, similar experiments testing spontaneous generation had yielded variable outcomes, a fact that proponents frequently cited as evidence that life arose spontaneously rather than by contamination.
Because Pasteur was focused on the cause, not the outcome, he thought of a way to address this too. If particles from the air were responsible, then differences in air should lead to differences in results. Pasteur wrote about the dust particles we can see in a shaft of light and how their abundance changes, or if windows are open or closed. These small environmental differences that could substantially alter what, and how many, particles are floating in the air. Armed with his new flasks, Pasteur went traveling to test them out in as many different ‘airs’ as he could manage. With windows opened or closed, in the rain or sunshine, in cities or countrysides, mountains or seasides. And apparently he even took them to a glacier.5 And across all of these varied conditions, his results were consistent. When airborne organisms are prevented from entering the broth, spoilage does not occur. And importantly, he was not stopping the air from entering altogether. The air itself was still getting through the flask necks, directly refuting one of the central arguments of spontaneous generation proponents.

A photomicrograph at 1000x magnification of Cronobacter sakazakii bacteria. Note the flagella.
Centers for Disease Control.
The last piece of the puzzle was to find the culprits. Based on his earlier work distinguishing the different microorganisms responsible for wine spoilage, this step was likely the easiest part. When he added cotton to his swan neck, Pasteur could filter the incoming air and trap the microorganisms. When he examined the cotton under the microscope, he’d captured organisms similar to those in his spoiled wines, provided further evidence that these tiny living organisms were responsible for the contamination, not a spontaneous event from ‘vital air’.
Even today, it is striking how close the proponents and opponents of spontaneous generation were in their hypotheses. There was a vital life force in the air. It was simply far too small to see with the naked eye. For hundreds of years they debated this, so publicly that the church and government became involved to lay the question to rest.
In 1862, Louis Pasteur was awarded the Alhumbert Prize by the Paris Academy. What Pasteur’s experiments accomplished was not just the resolution of a philosophical question, but a shift in how we understand life itself. These microbes were not rare or exceptional. They were all around us. Pasteur went on to show that the same microorganisms responsible for spoilage could also cause disease, what would later become known as ‘germ theory’. From there, he demonstrated that disease could be prevented. He developed the first laboratory‑based vaccines by weakening pathogens so they could train the body’s immune defenses without causing illness. Pasteur also advocated for practical changes that would profoundly impact our lives: personal hygiene to limit microbial spread, he developed sterilization techniques for surgical wounds and instruments. And of course, he patented his pasteurization process that helped beer and wine makers produce more consistent, unspoiled libations. In doing so, he laid the foundation for modern microbiology, medicine, and food safety.
Though the Alhumbert Prize ’ended’ the debate in official circles, scientific consensus is rarely instantaneous. Despite these observations in the 1860s, spontaneous generation arguments continued behind the scenes. It would be another decade or more before many researchers accepted that living organisms, rather than an elusive chemical process, were responsible for fermentation and spoilage.
“in the gap between what we can see and the invisible actors that do the work, uncertainty can build: an area ripe for misinformation and doubt.”
So, what finally ended the debate? The scientific process. Others repeated Pasteur’s experiments after making their own swan-necked flasks. And as more and more scientists reported similar results, the theory of spontaneous generation lost support. Further advances in tools like microscopy made it easier to observe these microorganisms. And as more people observed the culprits, germ theory gained a foothold. Germ theory not only explained the spoilage but also infection, contagion, and disease. Eventually, support for spontaneous generation faded altogether. Not because a prize represented a decisive answer, but because spontaneous generation no longer explained our experiences as well as germ theory.
When the science itself isn’t visible
Some may look back on the centuries of spontaneous generation debate as a relic of a less-informed time and people. But those people were not wrong, there were simply limits to what they could see and measure. And things we cannot see often create fear, especially when we can clearly see their effects. What ultimately resolved the debate over these unseen organisms was not a single epiphany, but the diligent work of scientists over generations, developing new ways to observe, test, and trust what they could not see. To trust what lay beyond the naked eye. This process took time, repetition, and a little creativity.
Today we face similar tensions. We expect science to deliver clear, certain answers very quickly. Answers that are reported in the news, seemingly in real time. But the history of discovery teaches us differently: scientific progress is often slow, uneven, and dependent on the tools we have available. Of course science today moves at a much faster pace than 200 years ago. And this is largely due to the incredible knowledge, tools, and methods that have been developed over centuries. But while science is still developing, disagreement is not a failure of science, it is a part of the process.
We saw this play out in real time with the development of the COVID-19 vaccines. In the face of this global crisis, people were looking for a cure, any wisp of hope that could end the nightmare: a quick and speedy cure. And in this case, science could deliver. So many news stories reported on the ‘radical new research’ being put to the test for the first time. A specific and incredibly effective vaccine in record time. A miracle cure!
What was less reported were the dozens of scientists who had worked for decades on the technologies that made these rapid vaccines possible: the immunology, the mRNA platforms, the delivery systems, even the sequencing. And we can see this throughout history: the discoveries we now attribute to a single name were actually shaped by many hands. But the public did not see the careful, incremental, and repeatedly tested science that these vaccines were built on. They saw an untested miracle. And in that gap between what we can see and the invisible actors that do the work, uncertainty can build: an area ripe for misinformation and doubt.
This gap, where uncertainty takes hold, was not new in 2020. It had been building for some time, and in recent years, it has only widened in recent years. We still distrust that which we cannot see. And in that space, we have become selective in what we choose to trust. We readily accept some truths about the invisible world. Many people would balk at the idea of not sterilizing equipment before their surgery, and even embrace the idea that our gut microbiome can support better health. Yet these same people are hesitant to believe that bacteria can make us dangerously sick when they appear in raw milk.
Why do we trust some truths about microbes but not others? Perhaps it’s because we can no longer see the illnesses that once defined everyday life. We don’t see polio much anymore, so we have forgotten its ravages. We’ve forgotten that less than 200 years ago, when Pasteur was seeking these same answers, nearly half of all children died before the age of five. And so, once again, we’re left trying to understand the things that we cannot see.
References
- Gottdenker P. Francesco Redi and the fly experiments. Bull Hist Med. 1979 Winter;53(4):575-92. PMID: 397843.
- Science Friday, July 15th, 2019. Interview with James Strick, Professor of Science, Technology and Society at Franklin and Marshall College in Lancaster, Pennsylvania.
- Adler R. Pasteur Begins Developing Germ Theory and Microbiology. Published online 2023.
- Cavaillon JM, Legout S. Louis Pasteur: Between Myth and Reality. Biomolecules. 2022 Apr 18;12(4):596. doi: 10.3390/biom12040596. PMID: 35454184.
- Dorit R. Winter 1859 – A single 30-day span begat much of modern biology. Scientific American, 2010 July-August;98(4):286.







